Interpretation of chemical formulae
that chemical compounds are substances made up of at least two chemical elements permanently bonded with each other;
that there are covalent compounds made of molecules and that ionic compounds are made of ions;
that molecular formula describes composition of the compound (covalent substances) or the smallest set of ions (ionic compounds);
molecular formulae of the following substances: nitrogen, chlorine, hydrogen, water, carbon dioxide, hydrogen chloride, ammonia, sodium chloride, magnesium chloride, aluminium chloride;
how sodium, magnesium, aluminium, chloride and sulphide ions are formed.
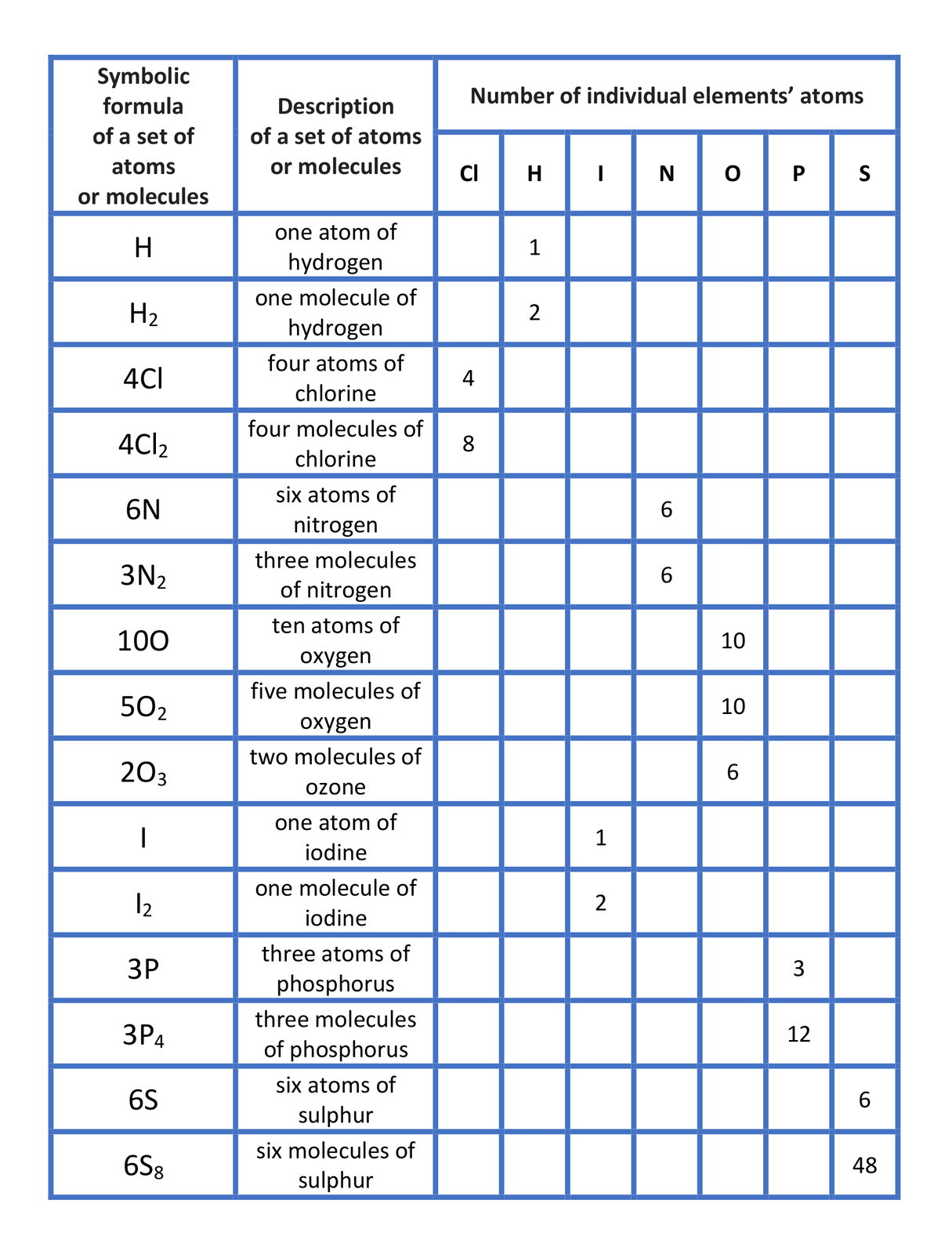
to explain how an atom differs from a molecule and indicate stoichiometric index and number in given chemical formula;
to interpret formulas such as: HIndeks dolny 22, 2H, 2HIndeks dolny 22;
to draw models based on formulae such as: HIndeks dolny 22, 2H, 2HIndeks dolny 22;
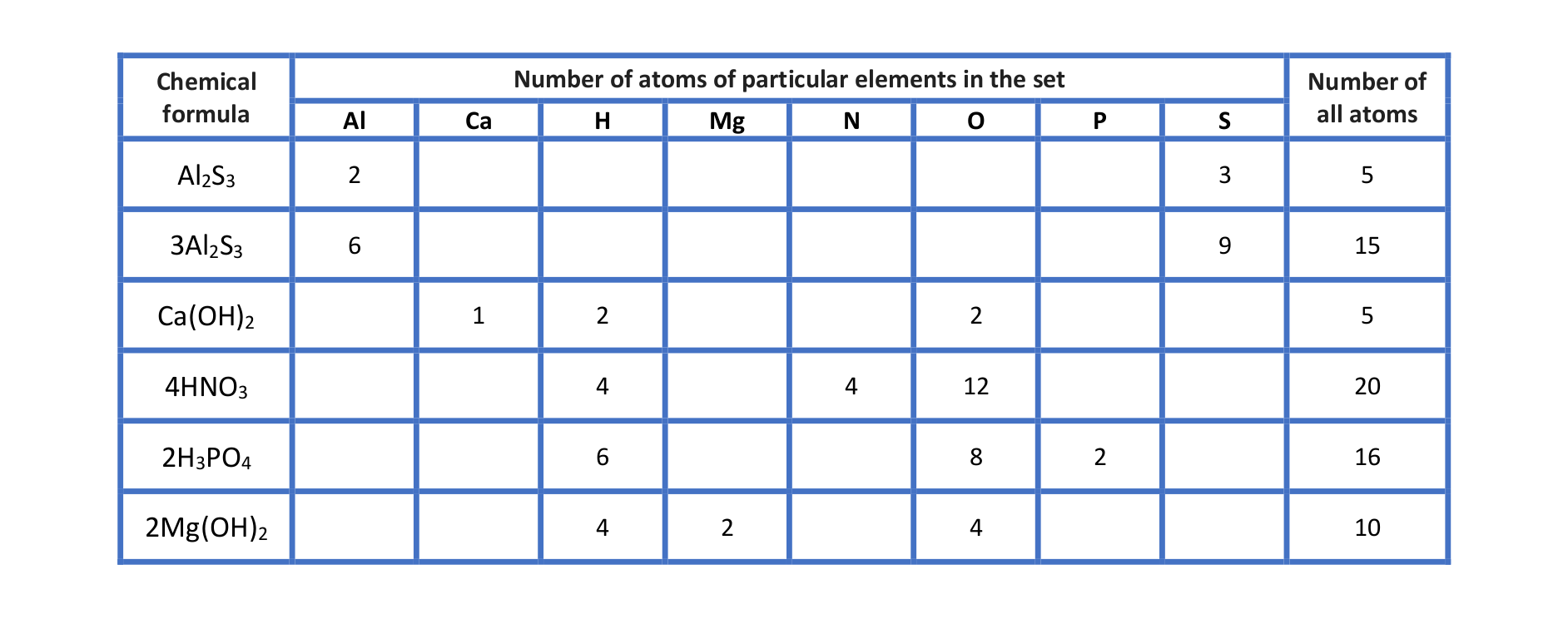
to specify the number of atoms in the given chemical formulae: HIndeks dolny 22O, 2HIndeks dolny 22O, AlIndeks dolny 22SIndeks dolny 33, 2AlIndeks dolny 22SIndeks dolny 33, HNOIndeks dolny 33, 3HNOIndeks dolny 33, HIndeks dolny 33POIndeks dolny 44, 2HIndeks dolny 33POIndeks dolny 44, Ca(OH)Indeks dolny 22, 3Ca(OH)Indeks dolny 22, CaIndeks dolny 33(POIndeks dolny 44)Indeks dolny 22, 2CaIndeks dolny 33(POIndeks dolny 44)Indeks dolny 22.
Nagranie dostępne na portalu epodreczniki.pl
Nagranie audio prezentujące structure of a covalent compound molecule.
The structure of a covalent compound molecule based on its formula
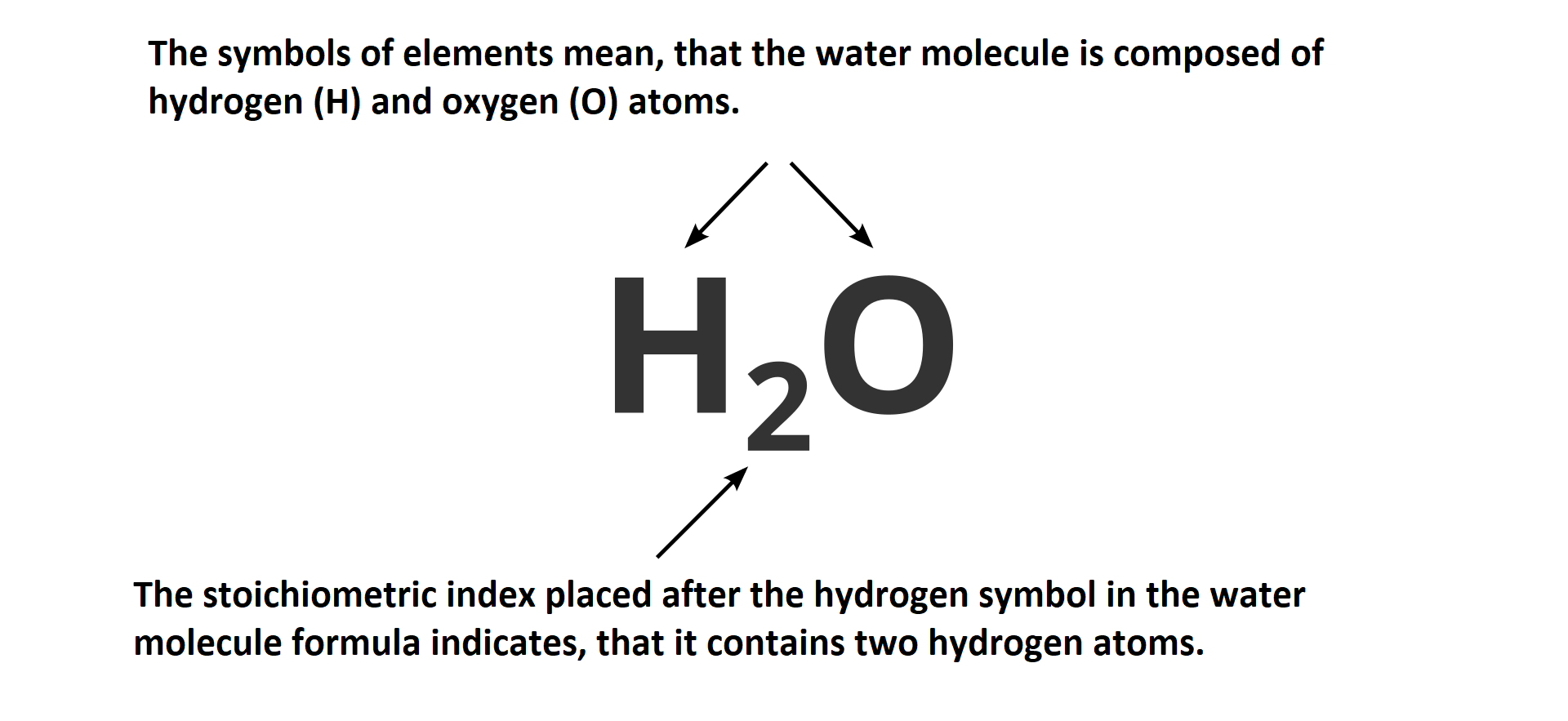
The chemical formula of a covalent compound describes the structure of its molecules. Symbols of elements used in the formulae inform about types of atoms creating a molecule and numbers placed on at the bottom right of the symbol inform about their number. The numbers we use to express the number of atoms in a molecule are called stoichiometric indicesstoichiometric indices. If stoichiometric index is equal to one, number 1 is not written down. In a chemical formulae of water stoichiometric index for hydrogen is 2 and the one for oxygen is – 1. On this basis, we can say that water molecule consists of two atoms of hydrogen and one atom of oxygen.
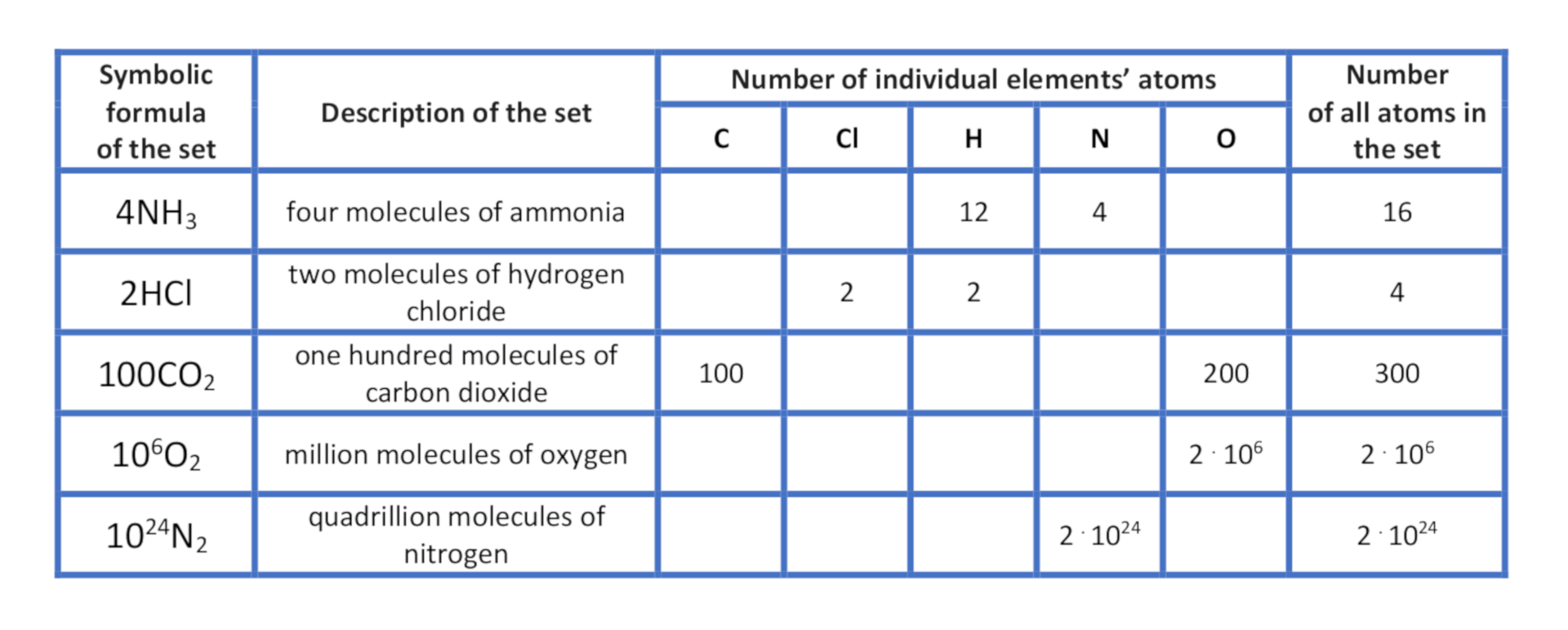
Description of the set of molecules
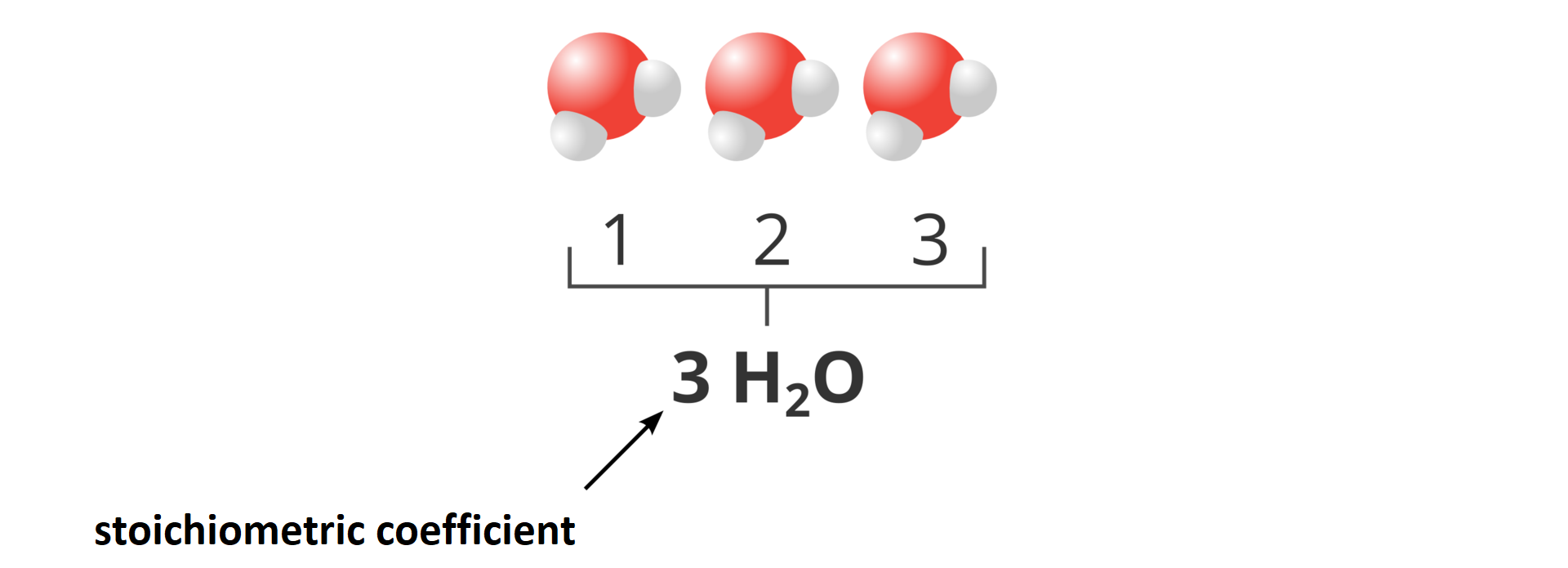
If we want to present a set consisting of more than one molecule, for example of three molecules of water, the number of these molecules is placed before its formula. Three molecules of water (, , ) will be described as . The number placed before the compound’s formulae is called stoichiometric numberstoichiometric number.
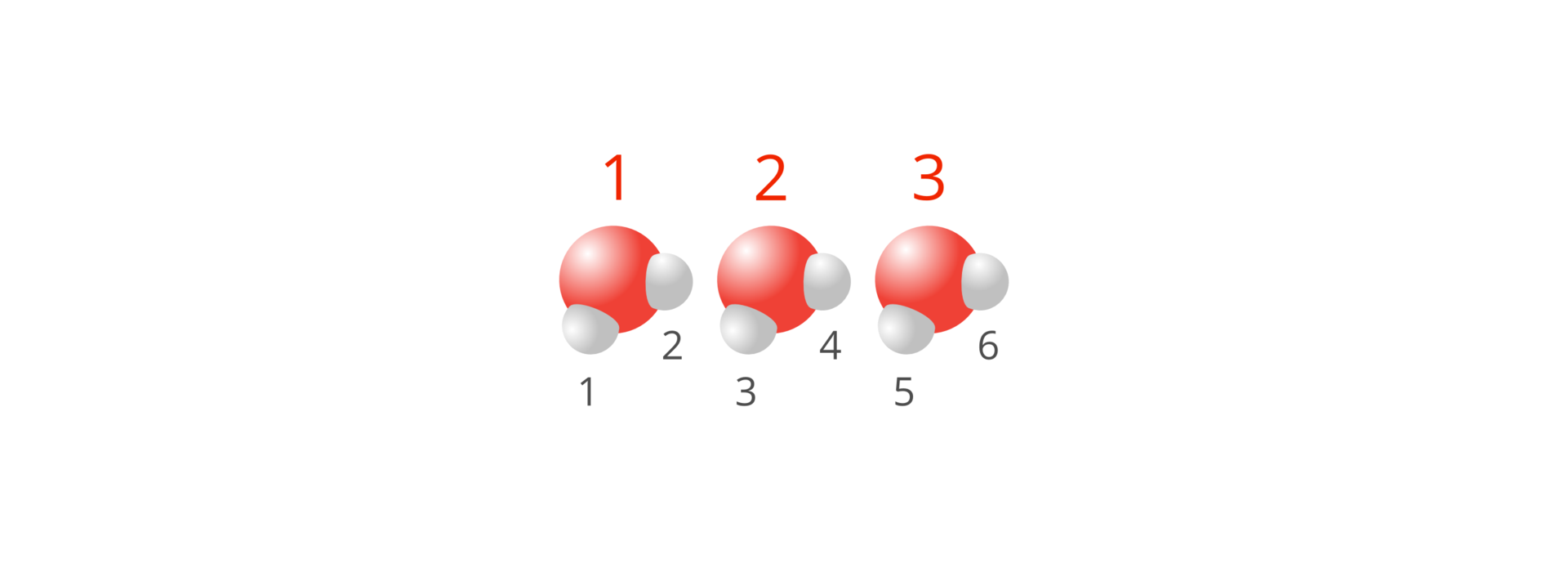
Let’s try to determine the number of individual atoms of elements in the set of three molecules of water: . One molecule contains two atoms of hydrogen and one atoms of oxygen, while three molecules contain in total , that is six atoms of hydrogen and , that is three atoms of oxygen.
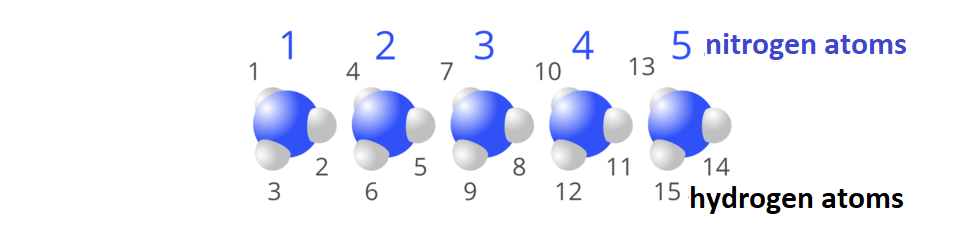
Five molecules of ammonia, , consist in total of () five atoms of nitrogen and () fifteen atoms of hydrogen.
How to describe molecules
In OIndeks dolny 33 molecule a stoichiometric index with an element’s symbol equal to 3 indicates that ozone molecules are made up of three atoms of oxygen.
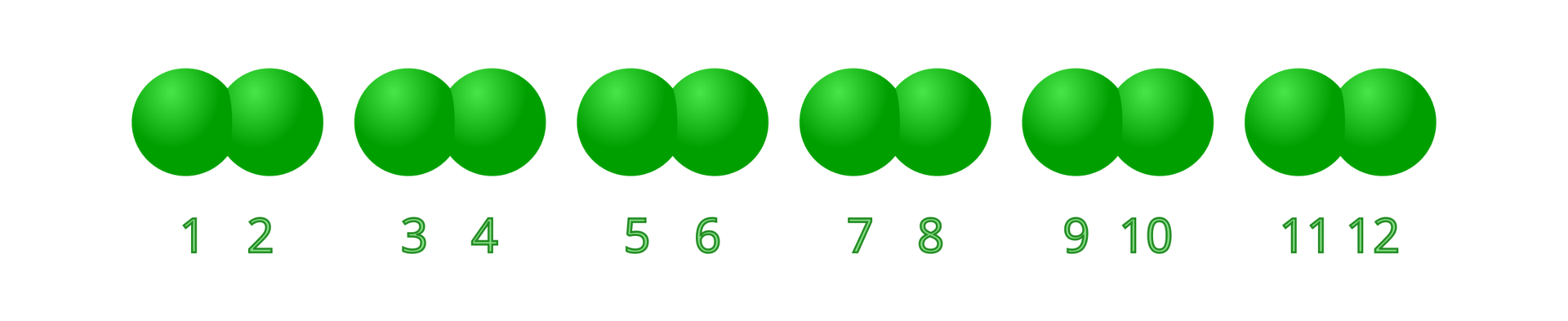
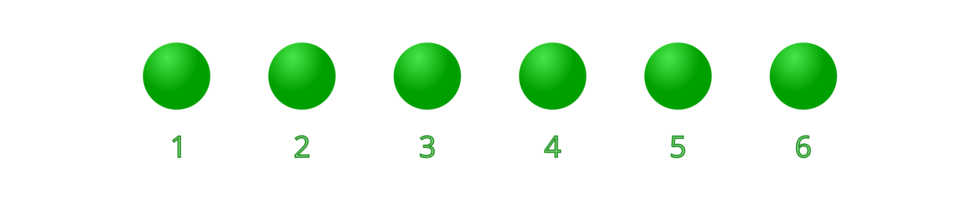
A stoichiometric number written before the symbol (for example – 6 atoms of chlorine) or a formula (for example – 6 molecules of chlorine) denotes the number of atoms or molecules. indicates 6 free unbound atoms of chlorine, while – indicates that there are 6 diatomic chlorine molecules, a total of 12 atoms.
Interpretation of formula of ionic compounds
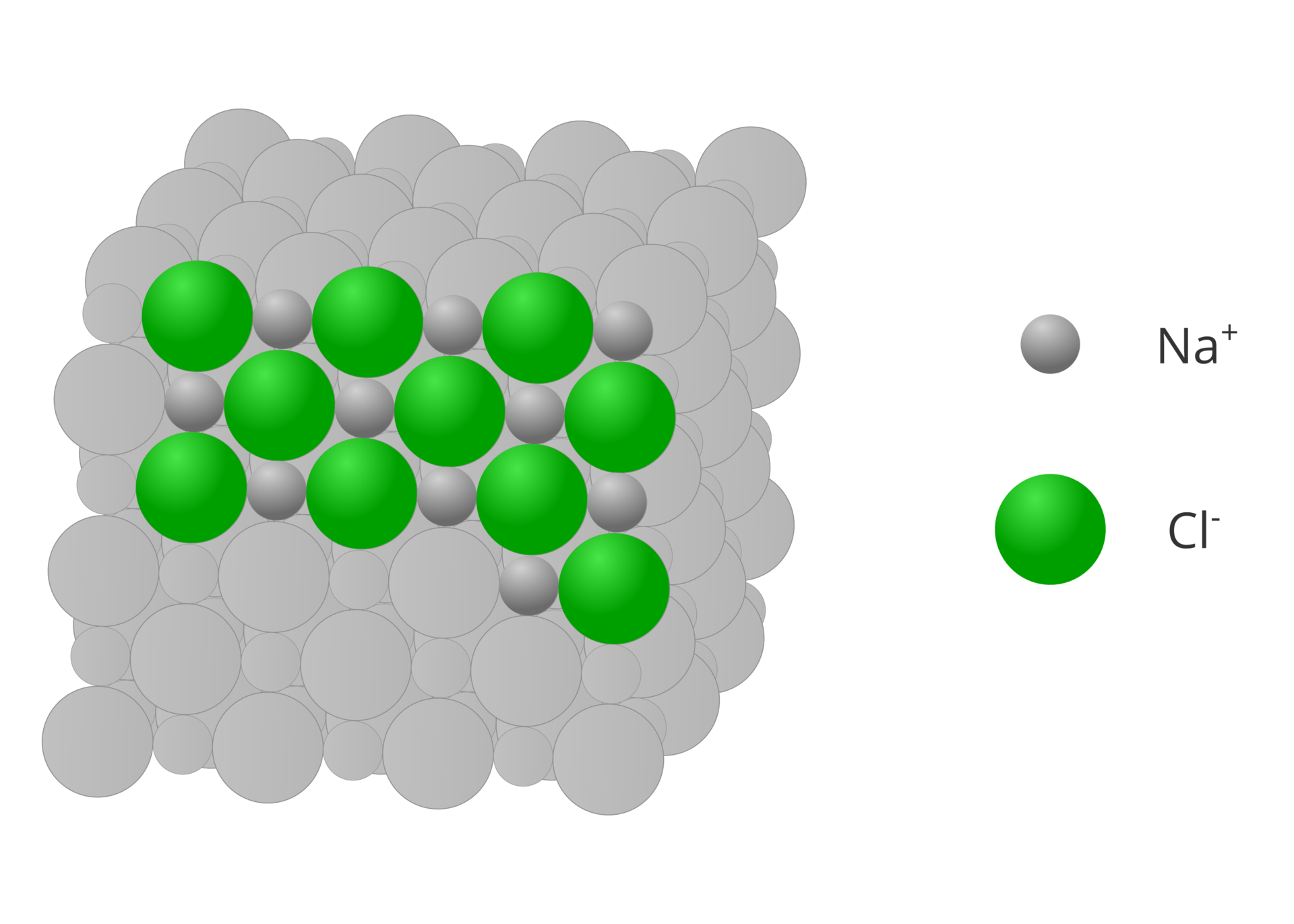
Ionic compounds are formed when metals bind with non‑metals. Such compounds are made of ions that are arranged alternately and form structures known as crystals. In a crystal there are no isolated groups of atoms that could be called molecules. A molecular formula of an ionic compound thus represents the smallest set of repetitive ions in a crystal. Stoichiometric indices denote a number of ions of given element in this set.
In ionic compounds consisting of two elements, metal is always a cation and non‑metal is – an anion. Sodium chloride is an example of such a compound. Its formula, , indicates that a crystal of sodium chloride is made of sodium cations () and chloride anions (), and their quantitative ratio is 1:1.
Numbers placed before the formula of an ionic compound (stoichiometric numbers) denote a number of the smallest sets making up for a crystal. For example denotes ten sets which consist of one sodium cation () and one chloride anion ().
Interpretation of chemical formulae
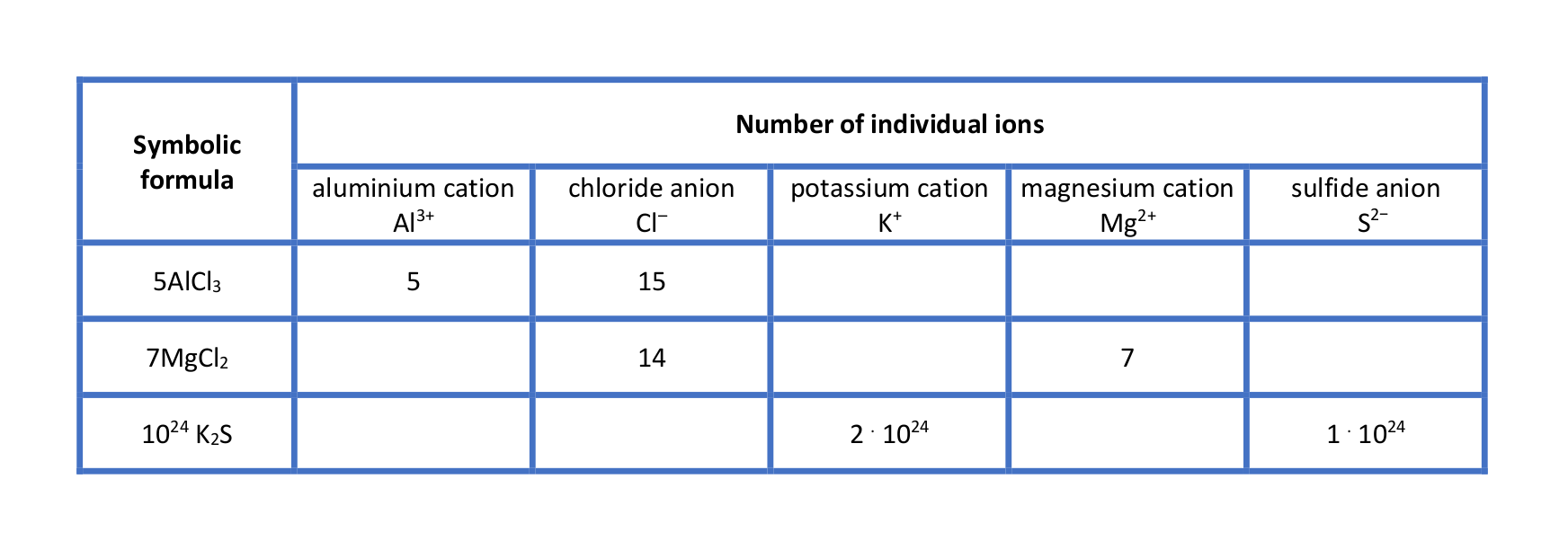
Have a look at the table “Interpretation of selected chemical formulae”. Analyse it together with your teacher and write down your conclusions.
Select an answer with correct number of atoms of individual elements in the following chemical formula: 3N2O5:
- 6 atoms of nitrogen and 15 atoms of oxygen
- 5 atoms of nitrogen and 8 atoms of oxygen
- 6 atoms of nitrogen and 8 atoms of oxygen
- 5 atoms of nitrogen and 15 atoms of oxygen
Select an answer with correct number of atoms of individual elements in the following chemical formula: 3Zn3(PO4)2:
- 9 atoms of zinc, 6 atoms of P and 24 atoms of oxygen
- 9 atoms of zinc, 6 atoms of P and 18 atoms of oxygen
- 6 atoms of zinc, 5 atoms of P and 9 atoms of oxygen
- 9 atoms of zinc, 5 atoms of P and 6 atoms of oxygen
Summary
A certain number of molecules, atoms and ions can be represented using chemical formulae.
The number of atoms in a molecule or ions in a unit is denoted using a stoichiometric index. In a formula this number is written down at the bottom right of the element’s symbol.
The number of atoms, molecules or units of an ionic compound is denoted using a stoichiometric number – the number written before the molecular formula of a compound.
Certain ionic substance has the following molecular formula: . Symbol denotes a metal and symbol – denotes a non‑metal. Which of the following ions could form an ionic compound? Write down the formula of this compound in your notebook.
Ions to choose from: , , , , .
Keywords
chemical formula, stoichiometric index, stoichiometric number, molecule, atom
Glossary
indeks stechiometryczny – liczba umieszczana u dołu z prawej strony symbolu pierwiastka we wzorze chemicznym substancji; jest ona równa liczbie atomów tego pierwiastka w cząsteczce lub w najmniejszym zbiorze powtarzających się jonów w krysztale (tzw. jednostce formalnej)
współczynnik stechiometryczny – liczba zapisywana przed symbolem lub wzorem chemicznym, która określa liczbę atomów, cząsteczek lub najmniejszych zbiorów powtarzających się jonów w krysztale