Octane number. Cracking and reforming.
that petrol is a mixture of hydrocarbons that consist of 5 to 12 carbon atoms in a molecule;
fuel for combustion engines is produced in fractional distillation of crude oil.
to explain what ON and octane number is;
to explain how to increase the value of octane number;
to explain practical significance of the octane number;
to explain how fuel quality can be improved and why it is done;
to define cracking and reforming;
to explain what cracking and reforming are in practice;
to justify the need to carry out cracking and reforming in the industry.
Petrol and gas oil
Cars with a combustion engine are powered by petrol and the ones with compression‑ignition (diesel) engine are powered by gas oil. What are the properties of these two types of fuels?
Formulate a research question and hypothesis before watching a video “Testing of the properties exhibited by petrol and gas oil”.

Film dostępny na portalu epodreczniki.pl
Na stole laboratoryjnym znajdują się 2 zlewki. W jednej jest bezbarwna ciecz (benzyna), a w drugiej ciecz bardziej żółtawa (olej napędowy). Eksperymentator podnosi obie zlewki i się przygląda przez chwilkę tym cieczom. Następnie naczynie z olejem napędowym odkłada i sprawdza zapach benzyny, wąchając opary znad tej cieczy w kierunku własnego nosa. Pierwszą zlewkę odkłada i bada zapach substancji w zlewce drugiej. Do jednej parownicy eksperymentator nalewa niewielką ilość benzyny silnikowej, zapala łuczywo i ostrożnie przykłada także palące się łuczywo do powierzchni benzyny. Benzyna natychmiast się zapala. Płomień jest jasny. Następnie eksperymentator do drugiej parownicy nalewa olej napędowy , zapala łuczywo i ostrożnie przykłada do powierzchni tego paliwa . Olej napędowy zapala się również, ale nieco wolniej i płomień jest żółty, nieco kopcący. Eksperymentator do jednego cylindra miarowego nalewa benzyny, a do drugiego oleju napędowego. Do obu cylindrów wprowadza areometr i obserwuje do jakiego poziomu zanurzy się jego nóżka. Zbliżenie na odczyt. Eksperymentator odczytuje wartość gęstości w miejscu, gdzie poziom cieczy przecina skalę areometru. Dla benzyny wartość powinna mieścić się w przedziale 0,720 – 0,775 g/cm3, a dla oleju napędowego 0.82 – 0.845 g/cm3. Do dwóch probówek należy nalać po 2 cm3 benzyny samochodowej. Do pierwszej dodać około 1 cm3 wody, a do drugiej około 1 cm3 oleju roślinnego. Zawartość probówek wymieszać wstrząsając. W przypadku mieszaniny wody z benzyną chemik pokazuje probówkę, zbliżenie kamery i widać wyraźnie 2 niemieszające się ciecze.(warstwa dolna o dwa razy mniejszej objętości to woda, a warstwa górna lekko żółta to benzyna). W drugiej probówce po wymieszaniu powstaje mętna prawie bezbarwna ciecz, co można zauważyć po zbliżeniu kamery na probówkę. Tę samą czynność należy powtórzyć z olejem napędowym. Podobne wyniki, z tym że warstwa oleju napędowego będzie bardziej żółta od benzyny.
Determining of combustibility and solubility of typical motor fuels in polar and non‑polar solvent.
Petrol and gas oil are flammable liquids that are soluble in certain solvents.
motor petrol,
gas oil,
areometer,
2 evaporating dishes,
4 test tubes,
2 measuring cylinders,
water,
vegetable oil,
torch.
The two beakers contain petrol and gas oil. Pay your attention to their state of aggregation, colour and odour.
Pour a small amount of petrol into one evaporating dish and carefully move brining torch to its surface. (This part of experiment is done by a teacher).
Pour a small amount of gas oil into the second evaporating dish and carefully move brining torch to its surface. (This part of experiment is done by a teacher).
Pour petrol to one measuring cylinder and gas oil to the other. Measure density of both fuels using areometer.
Pour about 2 cmIndeks górny 33 of petrol to two test tubes. Add about 1 cmIndeks górny 33 of water to thee first one and about 1 cmIndeks górny 33 of vegetable oil to the second one.
Repeat the same steps using gas oil.
Mix the contents of test tubes.
Cracking
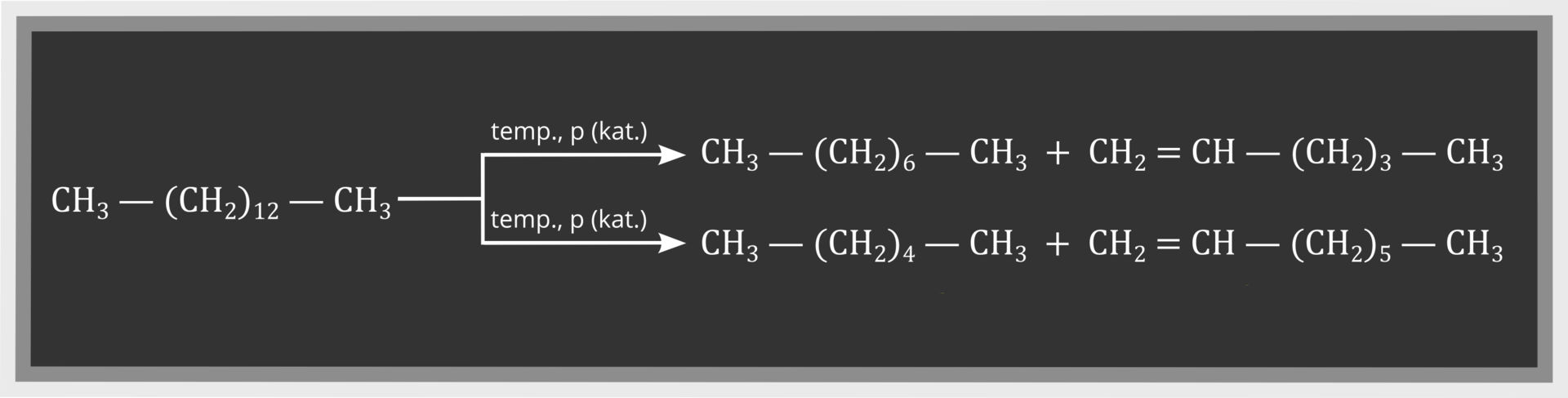
The quantity of petrol obtained directly as a result of fractional distillation of crude oil is not sufficient to meet the demand for this fuel. That it why it is produced in the processing of hard coal or synthesis gas, or in crackingcracking from the highest fractions obtained during fractional distillation of crude oil. Cracking (crack means break, burst) is a process that involves breakdown of long carbon chains into shorter ones. If cracking of long chains takes place under appropriate pressure (4–6 MPa) and temperature (740‑810 K), without air access, we are dealing with thermal cracking. If molecules with shorter chains are formed as a result of heating (at temp. of about 720 K), under the influence of elevated pressure and in the presence of a catalyst (for example AlClIndeks dolny 33, CrIndeks dolny 22OIndeks dolny 33, aluminosilicates), we are dealing with catalytic cracking. Catalyst makes it possible to decrease temperature and pressure needed during this process. Alkanes and alkenes with shorter carbon chain are formed as a result of cracking of alkanes, for example:
Octane number
Petrol obtained in the fractional distillation of crude oil is a mixture of hydrocarbons with simple carbon chains, which burns explosively, which is why it cannot be used as a fuel in vehicles. The octane number (ON) is the most significant parameter determining the quality of petrol. It reflects capacity of combustion in a cylinder of combustion engine without detonating (igniting). A mixture of petrol and air ignited by a sparking plug should burn evenly and not too quickly, because too rapid combustion causes uneven engine operation and its premature wear. Patrol of ON = 98 burns like a mixture consisting of 98% of isooctane and 2% of n‑heptane (n‑alkanes have the lowest octane numbers).
Antidetonators (anti‑knock agents)
The octane number of petrol can be increased using the so‑called antidetonatorsantidetonators, which are chemical anti‑knock agents. Iodine and aniline were one of the first of such agents. Employees of General Motors were testing 33,000 different chemical substances within 7 years and in 1922 they discovered and patented the anti‑knock effect of tetraethyllead – a compound with the following formula: Pb(CIndeks dolny 22HIndeks dolny 55)Indeks dolny 44. This substance turned out to be cheap and very effective because ON of petrol was increased from 60 to 96 if merely 0.15 g of this reagent was added to 1 dmIndeks górny 33 of petrol. However, it was soon realised that highly toxic lead compounds are released into the atmosphere with exhaust gases, posing a threat to people and natural environment. Nowadays, lead‑free petrols are used with antidetonators, mainly ethers, such as methyl tert‑butyl ether (MTBE) and ethyl tert‑butyl ether (ETBE) or alcohols (for example methanol and ethanol).
Reforming
ReformingReforming is another process used to increase the octane number of petrol. It consists in changing the components of gasoline at high temperature and in presence of catalysis, as a result of which branched and cyclic hydrocarbons (including aromatic ones) are formed. When these hydrocarbons are added to fuel, they increase its octane number. Reforming leads to: isomerization (production of hydrocarbons with the same composition but different structure), cyclization (conversion of chain hydrocarbons into ring hydrocarbons) and aromatization (transformation of chain hydrocarbons into ring hydrocarbons with a specific structure; those who will have extended chemistry curriculum will learn more about these hydrocarbons).
Mark true statements.
- Nowadays tetraethyllead Pb(C2H5)4 is used as an anti-knock agent (commonly known as antidetonator).
- Octane number is a parameter that measures resistance of petrol to knocking.
- Petrol is a mixture of hydrocarbons that consist of 7 to 15 carbon atoms in a molecule.
- Cracking is the breakdown of a large alkane into smaller molecules.
- Petrol is used to disinfect, remove fats and dressing residue from skin.
Create a multiple-choice test based on today's lesson. Then exchange your questions with a friend or classmate.
Question: ...
- ...
- ...
- ...
- ...
Summary
Petrol is obtained in the fractional distillation of crude oil and needs to be modified before it can become a motor fuel.
Octane number is a parameter that measures resistance of petrol to knocking.
Antidetonators are anti‑knock agents, which increase the octane number of petrol.
Cracking is a method for obtaining petrol from heavier fractions of crude oil distillation.
Reforming is a methods for obtaining high‑octane components of petrol (branched and ring hydrocarbons) from simple hydrocarbon.
Keywords
antidetonators, cracking, reforming
Glossary
antydetonatory - substancje przeciwstukowe dodawane do benzyny w celu podwyższenia jej liczby oktanowej
liczba oktanowa - parametr będący miarą odporności benzyny na spalanie detonacyjne (spalanie stukowe)
kraking - proces rozpadu alkanów o długich łańcuchach węglowych na związki o łańcuchach zawierających mniejszą liczbę atomów węgla
reforming - proces przekształcania węglowodorów o prostych łańcuchach węglowych w węglowodory o łańcuchach rozgałęzionych lub węglowodory pierścieniowe