Soap in the fight for cleanliness - dirt removal mechanism
what are characteristics of polar and nonpolar substances;
what are the properties of water which is a polar solvent;
what physical and chemical properties have fats;
what are the properties of hydroxides and what are characteristics of their aqueous solutions.
on the basis of the diagram, to discuss the mechanism of dirt removal;
to explain the process of removing dirt with the use of soap;
how to test the effect of water hardness on the formation of sparingly soluble compounds;
to describe how the hardness of water affects the washing process.
Does the addition of soap to the mixture of water and oil affect the process of their mixing?
The addition of soap to water and oil will allow mixing of the two immiscible liquids, i.e. dispersion of oil particles in water.
soap solution,
oil,
water,
2 tubes.
Add a small amount of oil to the test tube and shake it.
Observe the changes.
Add a similar amount of oil to the second tube containing the soap solution.
Shake the contents of the test tube and compare the effect of this test with the previous sample.
During the experiment, it can be observed that the soap affects the mixing of water and oil (polar and nonpolar liquid).
Watch the movie summarizing the experiment. Consider how the soap allows you to get rid of dirt consisting of from fat?

Film dostępny na portalu epodreczniki.pl
Film pokazuje eksperyment. Dwie probówki: w jednej woda, w drugiej roztwór mydła. Do wody dodajemy olej. Zatykamy korkiem probówkę. Przechylamy probówkę w celu wymieszania. Olej nie miesza się z wodą – unosi się na jej powierzchni. Do drugiej probówki z mydłem również dodajemy olej. Po wymieszaniu w probówce tworzy się piana.
Adding soap to the water during washing reduces its surface tension. In the presence of soap, water moistens the hydrophobic surfaces better, thus the removal of dirt is easier. Soap supports the removal of dirt, interacting with its particles, which along with soapy foam float on the surface of the water.
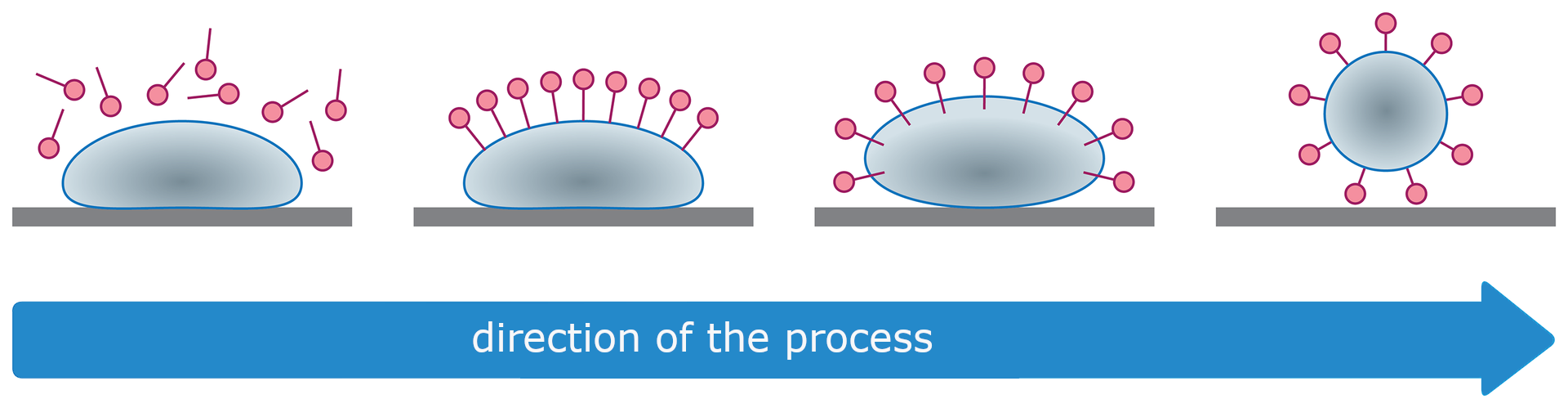
It happens that during washing a floccose precipitate forms in the water. It happens when the water used for washing is hard.
Hard waterHard water contains significant amounts of calcium and magnesium salts dissolved in it. When cleaning and washing in hard water using soap, insoluble calcium and magnesium salts form as a precipitate. The soap used for washing does not foam, and the formation of foam is a necessary condition for removing dirt. Sample equations of reactions can be written as follows:
There are two types of water hardness - carbonate hardness (temporary) caused by the presence of calcium and magnesium bicarbonates, and permanent hardness, which is caused by other calcium and magnesium salts (chlorides, nitrates, sulphates) dissolved in water.
When water containing calcium and magnesium bicarbonates is boiled, reactions take place, as a result of which calcium carbonate and magnesium carbonate settle on the walls of the vessel (e.g. kettle) in the form of a solid, colloquially called fur. The process of removing temporary hardness, resulting from the presence of calcium ions in water, is described by the equation:
Calcium and magnesium ions that cause water hardness are removed. The water softening effect is obtained. Therefore, carbonate hardness is called temporary hardness. Water hardness is most often expressed in German or French degrees.
German degree [°n] is equal to the amount of salts contained in water, which is chemically equivalent to 10 mg CaO.
French degree [°f] is equal to the amount of salts contained in water, which is chemically equivalent to 10 mg CaCOIndeks dolny 33.
[°n] | Water hardness |
0–4 | very soft water |
4–8 | soft water |
8–12 | moderately hard water |
12–18 | quite hard water |
18–30 | hard water |
Higher than 30 | very hard water |
Manufacturers of washing machines and dishwashers often warn against the action of hard water on these devices and recommend – to protect them from scaling – using water softeners. Information whether the water we have in the tap is hard is also important for aquarium fish farmers and users of water filters. Currently, products for testing the hardness of water at home are available.
Watch the experiment "Testing the behavior of a mixture of water and oil in the presence of soap" to select the correct answers.
- No foam is formed in the first beaker.
- Insoluble soap is formed in the second beaker.
- Foam is formed in the first beaker.
- Foam is formed in the second beaker.
Complete the sentences by choosing the appropriate phrases placed under the text.
air bubbles, floats, reduction, dirt particle, ions, dirt particles, micelles, foam, it melts under, increase
If we add surfactants, such as dishwashing soap, to the water, it causes a ............................ of its surface tension. Then cations and anions of surfactants, e.g. soap, shampoo, washing powder, form in the water clusters called ............................, and this contributes to the fact that ............................ are closed inside of them. In this way, the light micelle ............................ on the surface of the water and that is how ............................ arises.
Summary
Soaps effectively remove dirt because they are surface‑active substances. Their specific structure - the presence of a hydrophilic head and a hydrophobic tail - reduces the surface tension of water.
Keywords
hard water, solvent, soft water
Glossary
woda twarda – woda zawierająca znaczne ilości rozpuszczonych w niej soli wapnia i magnezu.