Carbohydrates, fats, proteins, nucleic acids
matter is composed of various substances, e.g. water, salt, iron;
substances are composed of particles that give them characteristic properties;
organisms absorb substances from their surroundings and process them.
to give examples of elements which build organisms;
to explain why carbon and water are so important for living;
to present the connection between the chemical build and properties of carbohydrates, proteins, fats and nucleic acids and their biological functions.
Carbohydrates
CarbohydratesCarbohydrates, commonly known as sugars, are widespread organic chemical compoundschemical compounds that are composed of carbon, hydrogen and oxygen atoms. They include one molecule monosaccharides, disaccharides and sugars composed of long chains of monosaccharide units, which are called polysaccharides. Sugars with small particles are easily soluble in water, those with long particles – don’t dissolve easy. The first ones are sweet, others have no taste. Carbohydrates are for all beings the source of energy, they are also building material and supplementary material. Very significant are monosaccharides, such as glucoseglucose and fructose , which provide energy to cells, as well as ribose and deoxyribose – ingredients of nucleic acids. Disaccharides are: sucrose – used for example to sweeten the tea, lactose – sugar found in milk, maltose – present in sprouting cereals, in nectar and pollens. Polysaccharides are composed of few thousands of glucose particles connected with each other. These are: starchstarch, glycogenglycogen, cellulosecellulose and chitinchitin.
Arrange a crossword using information from the abstract. Present it to your friends to solve.
Explain why glycogen is stored in muscles.
Observation of the shape of the grains of starch.
raw potato,
small knife,
instruments for the microscope,
microscope.
Cut a potato and gather the liquid that appeared on the cut with a knife.
Put a drop of that liquid on the slide and add a drop of water. Cover the microscopic section with a cover glass.
Look at the section under the microscope.
Complete a drawing.
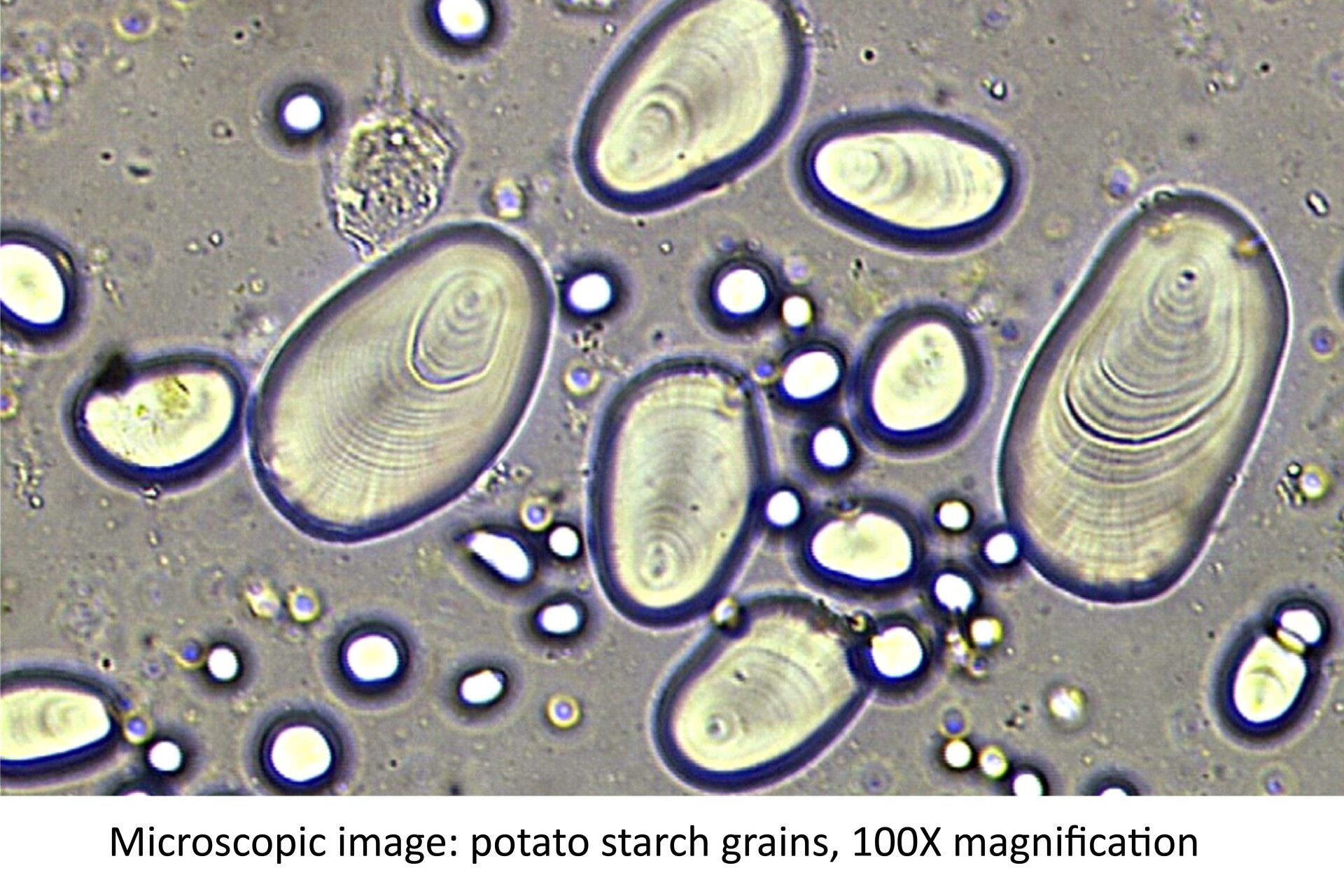
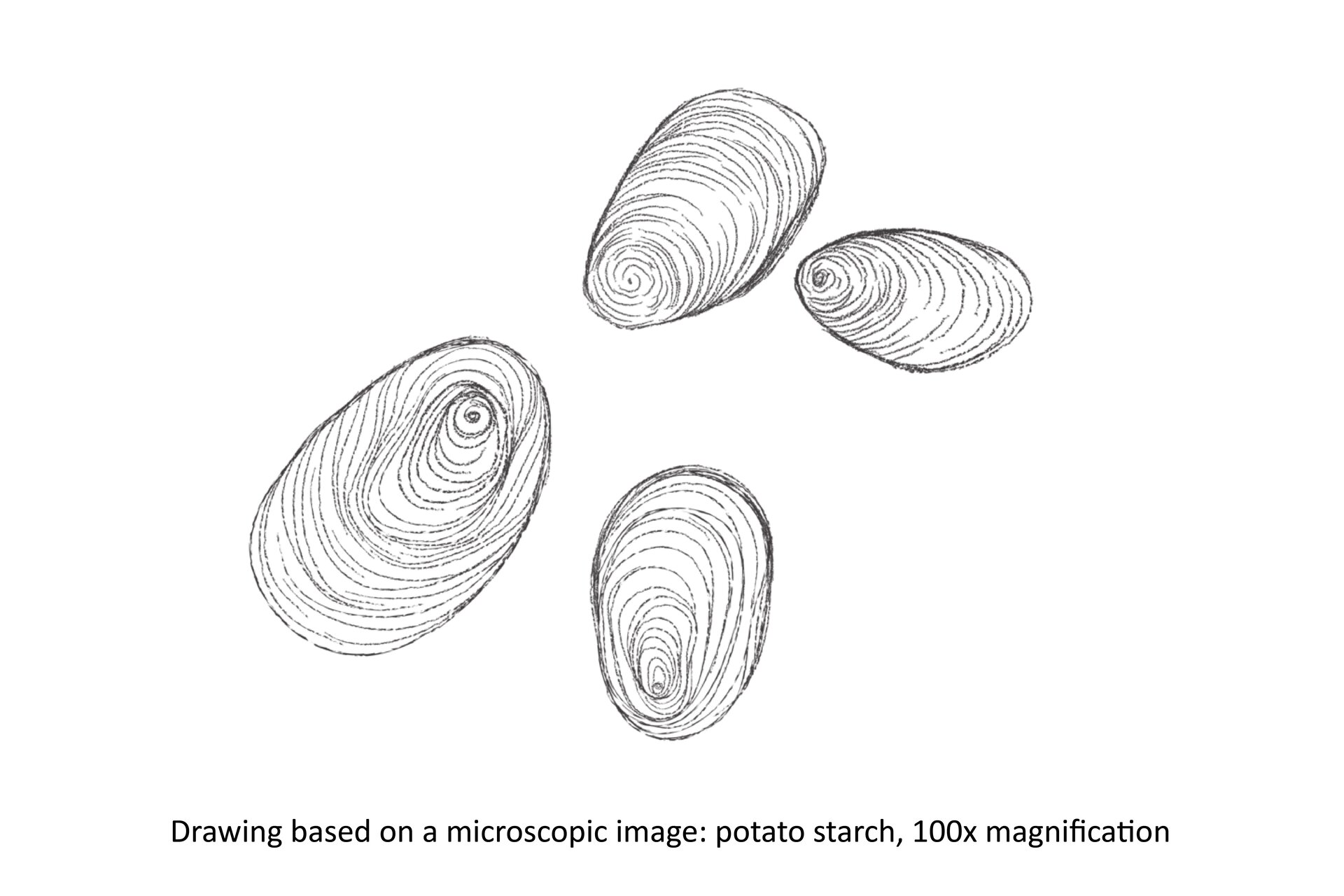
Grains of starch are oval.
Fats
FatsFats are composed of carbon, oxygen and hydrogen. These compounds are a perfect spare material, which is a reach source of energy for processes that take place in cells. In plants, fats are stored in the form of an oil, mainly in seeds and some fruit. Animal fats have solid form. They are mainly stored in fat tissue, but also in kidneys, liver, heart – internal organs vital for the body. They have protective and supportive functions there. A thick layer of fat tissue protects animals against mechanical injuries. It also protects them from low temperatures, being an ideal heat‑insulating material. Fat derivatives called phospholipids are very important ingredients that build cell membranes of all living organisms.
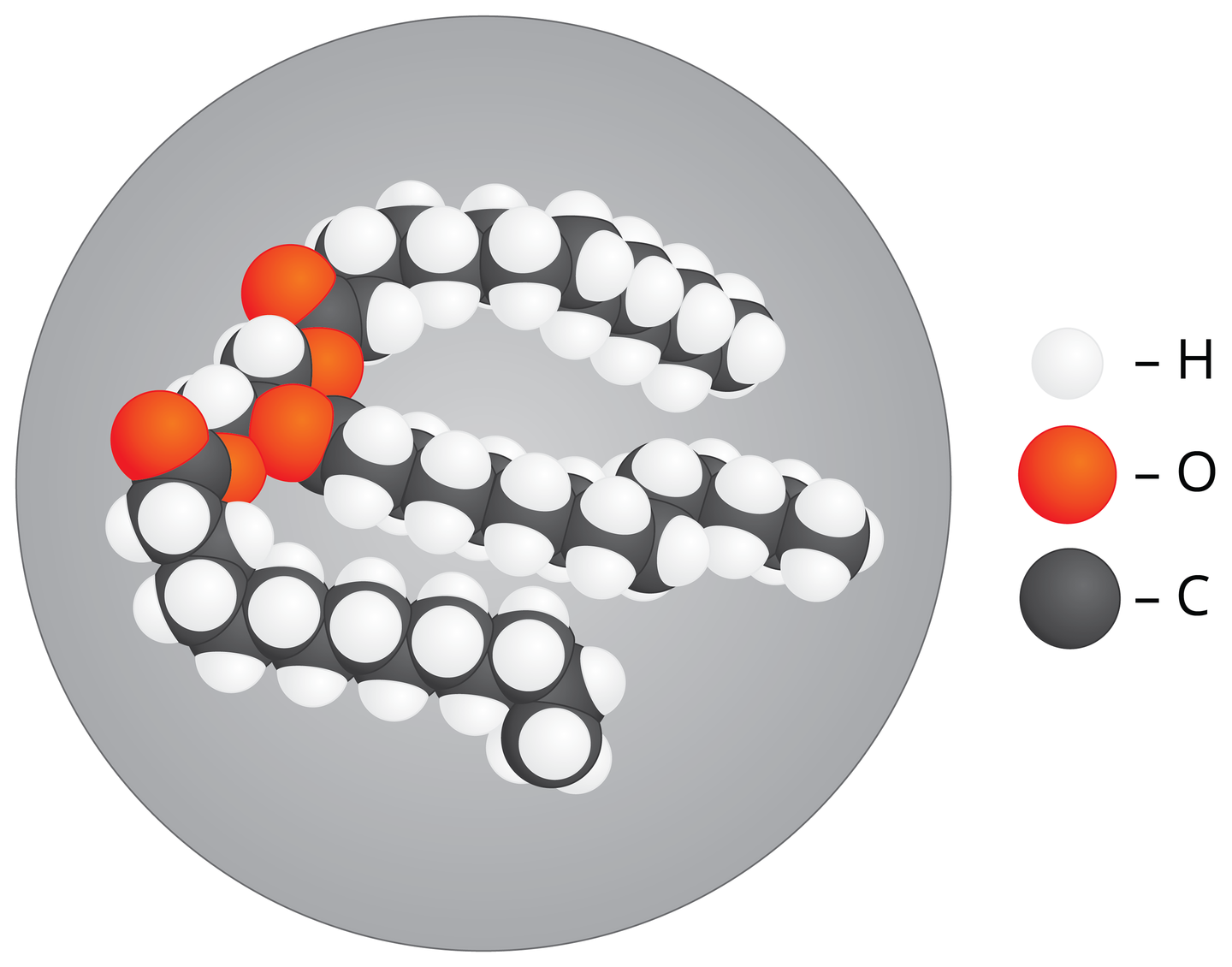
Plants store fats mainly in seeds. Give examples of plants that have the most fat.
Proteins
Proteins are organic compoundsorganic compounds which have elements such as carbon, hydrogen, oxygen, nitrogen and sulphur. They are composed of amino acids connected with each other and forming long chains. There are only 20 amino acids in nature, but there are tens of thousands of proteins. Each amino acid appears multiple times in a protein. The sequence of amino acids in a protein chain decides about the great variety of these compounds and about different functions of proteins. Building proteinsproteins are the ingredient of all organisms, they appear e.g. in cell membranes. Enzymatic proteins decide about biochemical reactions in organisms. Immunological proteins, also known as antibodies, fight against foreign bodies which penetrate our organisms and can cause infections or allergies. Growth hormone and insulin are examples of regulatory proteins that are responsible for regulating the functioning of our body. There are also proteins that allow us to contract muscles. Transporting proteins transport substances between cells. They can be spare material in plants. Especially rich in proteins are peas, beans, broad beans, soy and lentils. It happens very often that these compounds can be used as energy source. The properties of proteins depend on their complex spatial structure.
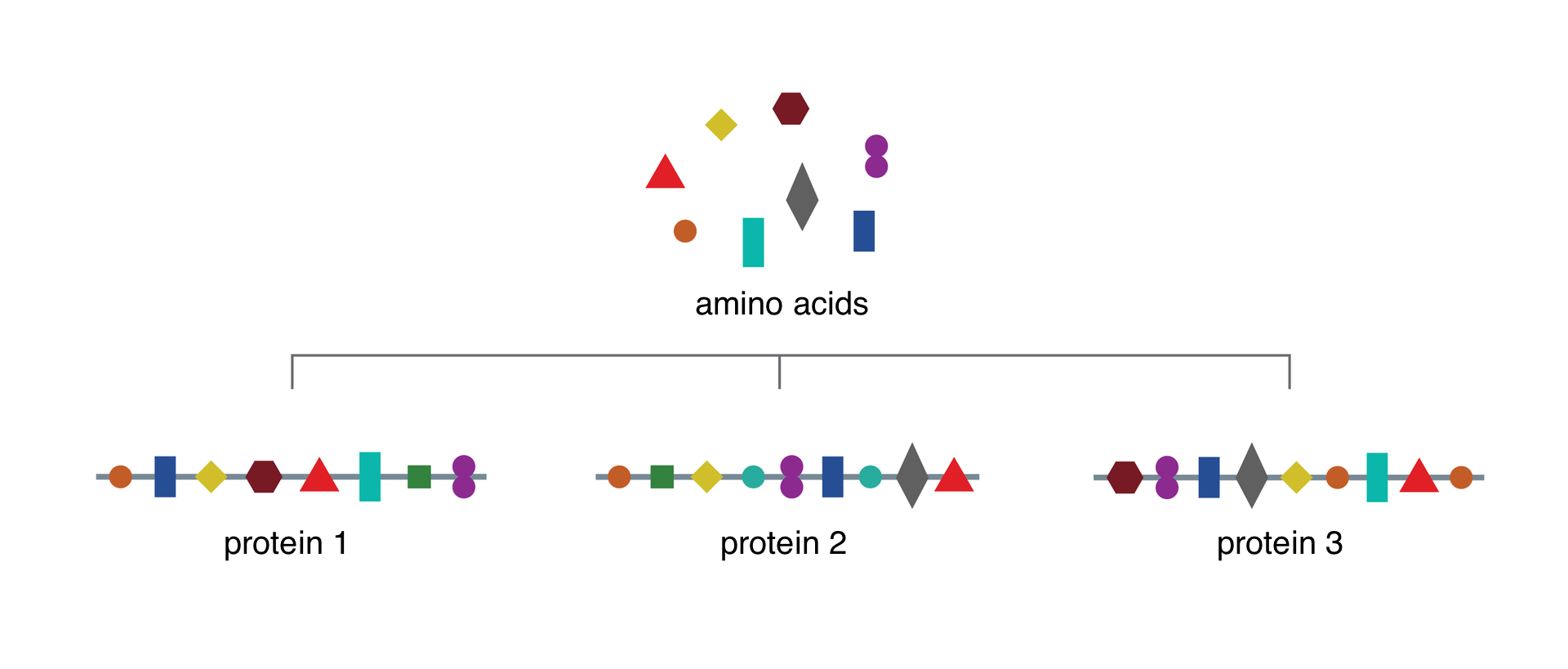
Explain why:
the scale of a thermometer used for measuring fever ends at 41°C;
prolonged high fever is dangerous for our health and must be lowered.
Nucleic acids
Nucleic acidsNucleic acids, which appear in all organisms and viruses, are organic compounds which contain genetic information, that is “the recipe for an organism”. Even though the amount of nucleic acids in cells is around 1%, their role in organisms is extremely important. Each representative of a given species has characteristic, one‑of‑a-kind genetic material, which heavily influences how that representative looks, develops and functions.
Nucleic acids particles have the form of chains that consist of thousands repeated variations of four basic units – nucleitides. Their order, just like the order of letters in a sentence, is the content of the “recipe for an organism”. Deoxyribonucleic acid (DNA) is composed of two strings twisted around each other.
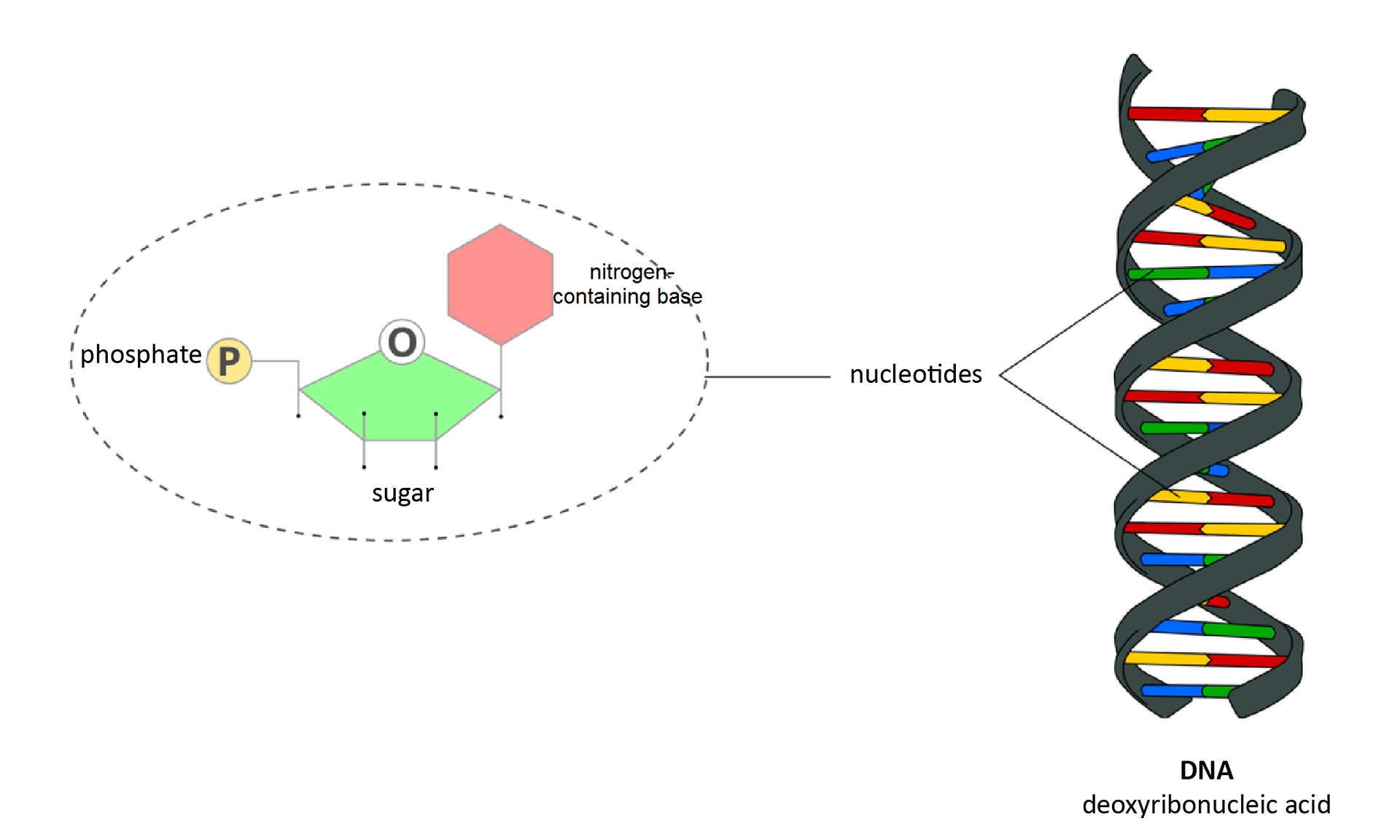
Explain why nucleic acids can be the source of energy for the organism. Justify your answer.
Mark two correct definitions regarding the functions of fats.
- They create an isolating layer that protects against the cold.
- They are the environment in which reactions in cells take place.
- They are the source of energy for the processes that take place in cells.
- They destroy microbes that penetrate the body.
The element which appears in all organic compounds is:
- calcium
- nitrogen
- carbon
- oxygen
Match the words and their definitions.
a chemical compound which has atoms of carbon that very often create long chains, and also atoms of other elements, polysaccharide that does not dissolve in water; it builds the cell walls of the plants, a compound which does not have atoms of carbon; the exceptions are carbon monoxides, carbonic acid and carbonates that include carbon, polysaccharide that does not dissolve in water; it builds the cell walls of the fungi and exoskeletons of the insects, multimolecular organic compounds that are composed of amino acids; they exist in all organisms, polysaccharide; it is a reserve source of energy for animals (appears mainly in liver and in muscles), polysaccharide composed of particles of glucose and does not dissolve in water; reserve material for plants, monosaccharide; basic source of energy for organisms, organic chemical compounds; they store genetic information of the body and act as intermediaries in protein production, a homogeneous substance that is composed of selected atoms permanently connected with each other to create particles
| proteins | |
| cellulose | |
| chitin | |
| glucose | |
| glycogen | |
| nucleic acids | |
| starch | |
| chemical compounds | |
| inorganic compound | |
| organic compound |
Summary
Organic compounds of organisms are proteins, fats, carbohydrates and nucleic acids.
Glucose is the main source of energy in cells.
Polysaccharides have building and spare functions.
Proteins are the basic building elements of cells and they regulate the life processes.
Fats are the source of energy and spare material for plants and animals.
DNA carries genetic information.
How was this lesson? Did you like it? Finish selected sentences.

Keywords
organic compounds, carbohydrates, proteins, fats, nucleic acids
Glossary
białka – wielkocząsteczkowe związki organiczne zbudowane z aminokwasów; występują we wszystkich organizmach; pełnią funkcje budulcowe, enzymatyczne, transportujące, odpornościowe i regulacyjne
celuloza – wielocukier nierozpuszczalny w wodzie; buduje ściany komórkowe roślin
chityna –wielocukier nierozpuszczalny w wodzie; buduje ściany komórkowe grzybów i szkielety zewnętrzne owadów
glukoza – cukier prosty; podstawowe źródło energii dla organizmów
glikogen – wielocukier; stanowi materiał zapasowy u zwierząt (występuje głównie w ich wątrobie i mięśniach)
kwasy nukleinowe – organiczne związki chemiczne; przechowują informację genetyczną organizmu i pośredniczą w produkcji białek; znane są dwa podstawowe typy naturalnych kwasów nukleinowych: kwasy deoksyrybonukleinowe (DNA) i kwasy rybonukleinowe (RNA)
skrobia – wielocukier zbudowany z cząsteczek glukozy, nierozpuszczalny w wodzie; materiał zapasowy roślin
tłuszcze – związki organiczne; stanowią substancje zapasowe roślin i zwierząt oraz materiał ochronny i termoizolacyjny u zwierząt
węglowodany – inaczej cukry; organiczne związki chemiczne złożone z atomów węgla oraz wodoru i tlenu; ze względu na budowę dzielone na cukry proste, dwucukry i wielocukry; jedna z podstawowych grup związków chemicznych wytwarzanych przez organizmy
związek chemiczny – jednorodna substancja złożona z wybranych atomów trwale połączonych w cząsteczki
związek organiczny – związek chemiczny, w skład którego wchodzą atomy węgla, często tworzące długie łańcuchy, a także atomy innych pierwiastków; wyjątkiem są zawierające węgiel tlenki węgla, kwas węglowy i węglany; związki organiczne powstają głównie w organizmach